|
Transfusion reactions are infrequent but can cause a patient’s condition to rapidly deteriorate. Any signs and symptoms suggesting a reaction should not be ignored, but rather assessed immediately. To minimize the risk of harm, early identification and prompt treatment are essential.
STOP!
- Stop the transfusion immediately
- Assess: rapid clinical assessment
- Check: confirm patient ID band matches blood swing label details
- Inspect: visual check of unit for turbidity, clots or abnormal appearance
- Talk with the Patient: establish status, inform and comfort
Recognise.
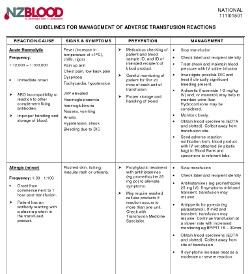
Signs and symptoms may include:
- Fever, chills, rigors
- Tachycardia, arrhythmias
- Hyper or hypotension, collapse
- Generalised flushing
- Rash, urticaria, angioedema
- Anxiety, severe apprehension
- Nausea, vomiting
- Pain (chest, loin, muscle, bone, abdominal,cannula site/vein)
- Dyspnoea, respiratory distress, hypoxia
- Pink/red/black urine or abnormal bleeding
Respond
Management and investigations
- If you suspect a potential acute transfusion reaction: STOP the transfusion immediately
- Undertake a rapid clinical assessment, check wristband ID to swing label ID, check unit and inform/comfort the patient
- Call for help
- Clinical Review/Treatment: see first response clinical algorithm
- Standard ATR Investigations - for ALL moderate and severe events are:
- EDTA (pink top) for serology - to Blood Bank
- Full blood count/film and U&E - to Pathology
- Ward urinalysis for blood/haemoglobin
- Additional Investigations: if...
? Haemolysis: consider haptoglobin, LDH, coag’s
? Respiratory Distress: consider CXR, ABGs, BNP
? Sepsis/Shock: consider blood cultures from patient
? Severe allergy/anaphylaxis: consider serum tryptase & query need for anti-IgA antibodies
- Further advice on management of ATRs is located here.
Report.
Haemovigilance to Blood Bank (BB)
- Report all mild, moderate and severe ATRs using the NZBS ATR Notification Form
- If the event is moderate or severe, remember to include the EDTA (pink) sample, the discontinued unit and blood IV infusion set with the ATR Form (111F009).
Advice on management, further transfusion needs or recurrent reactions should be discussed with the transfusion medicine specialist (TMS) or the
clinical haematology consultant.
Resources
More Info
|

Acute Transfusion Reaction (ATR) Notification Form

First Response Algorithm for the Management of Acute Transfusion Reactions (ATR)
Guidelines For Management Of Adverse Transfusion Reactions
|




