| ALERT |
Beriplex NZ is a four-factor prothrombin complex concentrate (4FPCC) – it is not interchangeable with 3FPCC (e.g. Prothrombinex-VF). |
|
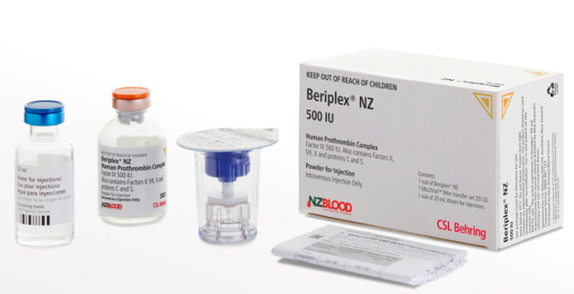
Presentation
|
- Beriplex NZ is a four factor prothrombin complex concentrate, supplied as a 500 IU vial containing factors II, VII, IX, and XIX, as well as Protein C and S (see datasheet for ranges of concentrations).
- Each single pack contains one vial of product, one 20 mL vial of Water for Injections and one Mix2Vial filter transfer set.
- Beriplex NZ also contains up to 343 mg (approximately 15 mmol) sodium and up to 200 IU heparin per 100 mL reconstituted solution.
|
|
|
ABO Compatibility
|
- A blood group is not required prior to release.
- Compatibility is not relevant for manufactured (fractionated) plasma product transfusion
|

Click for video
|
|
Storage and Reconstitution
|
- See the Mix2Vial System for intructions on reconsitution of the vial.
- Beriplex NZ is stored below 25°C in controlled Blood Bank conditions, and usually refrigerated at 2-8°C.
- Allow the vials to reach room temperature (up to 25°C) before reconstituting.
- Only remove the vials from the sealed box when ready to reconstitute (Beriplex NZ boxes are sealed to protect from light, product damage and tampering)
- Allow at least 10 minutes to reconstitute a vial of Beriplex NZ
- The technique for reconstituting Beriplex® products follows a similar, but slightly different sequence using the Mix2Vial system.
- Draw up reconstituted Beriplex NZ into a luer-lock syringe
- Do not dilute in saline or any other IV fluids or medications (the solution will not be stable and will precipitate)
- Once reconstituted infuse without delay as an IV bolus after completing all pre-blood checks at the bedside (including positive patient identification, product verification and the presence of signed informed consent)
- Multiple vials of the same product may be pooled together.
- For large volumes a syringe infusion pump can be used
- Do not refrigerate Beriplex NZ once it has been reconstituted.
|
|
Precautions
|
- Read manufacturer's instructions carefully.
- Note! Administering Beriplex NZ to patients who are not warfarinised substantially increases the risk of thromboembolism
- Always observe for turbidity / particulate material
(floaters).
- Pumps use is acceptable.
|
|
Rate
|
- Beriplex NZ should be given as a slow IV bolus injection into a peripheral or central venous access line.
- The rate must not exceed 0.12mL/kg/minute up to a maximum of at 8mL/minute.
- i.e. 3 IU / kg body weight/minute, up to a maximum of 210 IU /minute
- Administer without delay and within 3 hours of reconstituting (vials contain no antimicrobial additives)
- Some example weights and rates:
| Weight |
Rate |
| Over 65 kg |
Max rate: 8mL/min (480 mL/hour) or at least 2.5 minutes per 500 IU vial |
| 40-65 kg |
Max rate: 5ml/min (300 mL/hour)or at least 4 minutes per 500 IU vial |
| Under 40 kg |
Calculate at 0.12mL/kg/min or 7.2 ml/kg/hour |
|
|
Dose
|
- Beriplex NZ for reversal of warfarin: 15-50 IU/kg in the following situations:
- INR is ≥ 1.5 with life threatening (critical organ) bleeding
li>
- INR is ≥ 2.0 and there is clinically significant bleeding
- INR is ≥10.0 and the patient is at high risk of bleeding
- a pre-op patient where surgery is urgent, INR > 1.5 due to warfarin
- NOTE:In patients weighing more than 100kg, the dose of Beriplex NZ should be based on a capped 100kg body weight and maximum dose of 50 IU/kg (i.e. the absolute maximum dose is 5000 IU)
- NOTE:Repeat dosing is not supported by clinical data and the product information documents the risk of thrombosis with repeat dosing . If repeat dosing is required within 14 days, please discuss this witha NZBS Transfusion Medicine Specialist (TMS)
- NOTE:INR correction is commonly reached 30 minutes after Beriplex® NZ injection. The co-administration of vitamin K usually takes effect within 4-6 hours and is used for more sustained reversal
- THANZ warfarin reversal guidelines (2024)
|
|
Rivaroxaban guidance
|
- There is no specific agent for the reversal of rivaroxaban in New Zealand
- In the event of emergency surgery or major bleeding, Beriplex NZ may be considered as part of the management plan
|
|
Monitoring and Traceability
|
- Recipients should remain under observation for 20 minutes following administration in case they experience an immediate adverse event requiring treatment.
- Ensure the swing label from every box is attached to the local transfusion record as a permanent record
|
|
Compatibility with other medicines and fluids
|
- Beriplex® NZ cannot be co-mixed or co-administered with any IV fluid, blood product or medication
- Reconstitute with the supplied WFI only; never dilute in saline or other IV fluids or medications
- Do flush the IV access/line with saline prior and post the injection
|
|
More Info
|
|



